Other
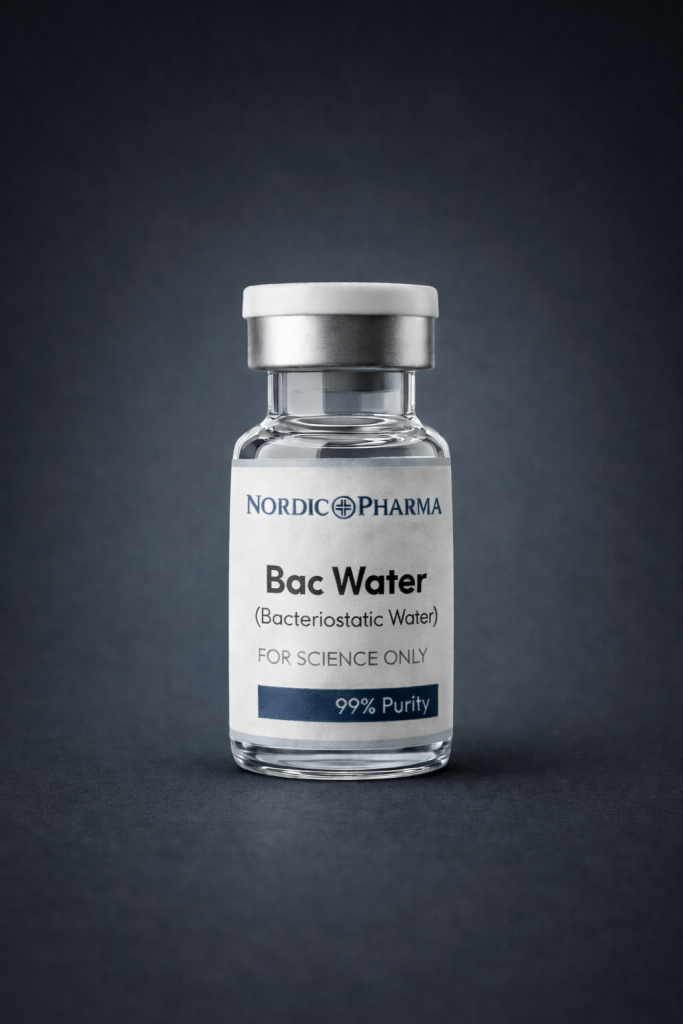
BAC Water | 10 vials (TRANSPORTATION ISSUES!)
Research Grade
10 vials
Price range: 29,99 € through 59,99 €
Bacteriostatic Water for Injection is a sterile, non-pyrogenic preparation of water for injection containing 0.9% (9 mg/mL) benzyl alcohol added as a bacteriostatic preservative . It is supplied in multi-dose containers, allowing repeated withdrawals to dilute or dissolve drugs for injection over an extended period .
Key Specifications:
| Property | Specification |
|---|---|
| Composition | Water for Injection (WFI) + 0.9% benzyl alcohol |
| pH Range | 4.5 to 7.0 |
| Format | Sterile liquid in multi-dose vials |
| Fill Volumes | 10 mL, 30 mL, and other sizes |
| Sterilization | 0.22 µm filtration, aseptically filled |
| Storage | Room temperature (15-25°C) |
| In-Use Period | Up to 28 days after first puncture (with proper aseptic technique) |
| Manufacturing | USP specifications; GMP/ISO facilities |
Primary Applications:
| Category | Applications |
|---|---|
| Peptide Reconstitution | Dissolving lyophilized peptides for research or clinical use |
| Multi-Dose Preparations | Allowing repeated withdrawals from a single vial without bacterial growth |
| Pharmaceutical Manufacturing | Diluting or dissolving drugs that require an aqueous vehicle before injection |
| Laboratory Research | Preparing working solutions from lyophilized proteins, antibodies, and reagents |
Important Notice:
FOR RESEARCH USE ONLY OR PRESCRIPTION USE ONLY.
Select Option & Quantity
Key Benefits
- High purity research compound (>98%)
- Third-party tested and verified
- Lyophilized for maximum stability
- Certificate of analysis available